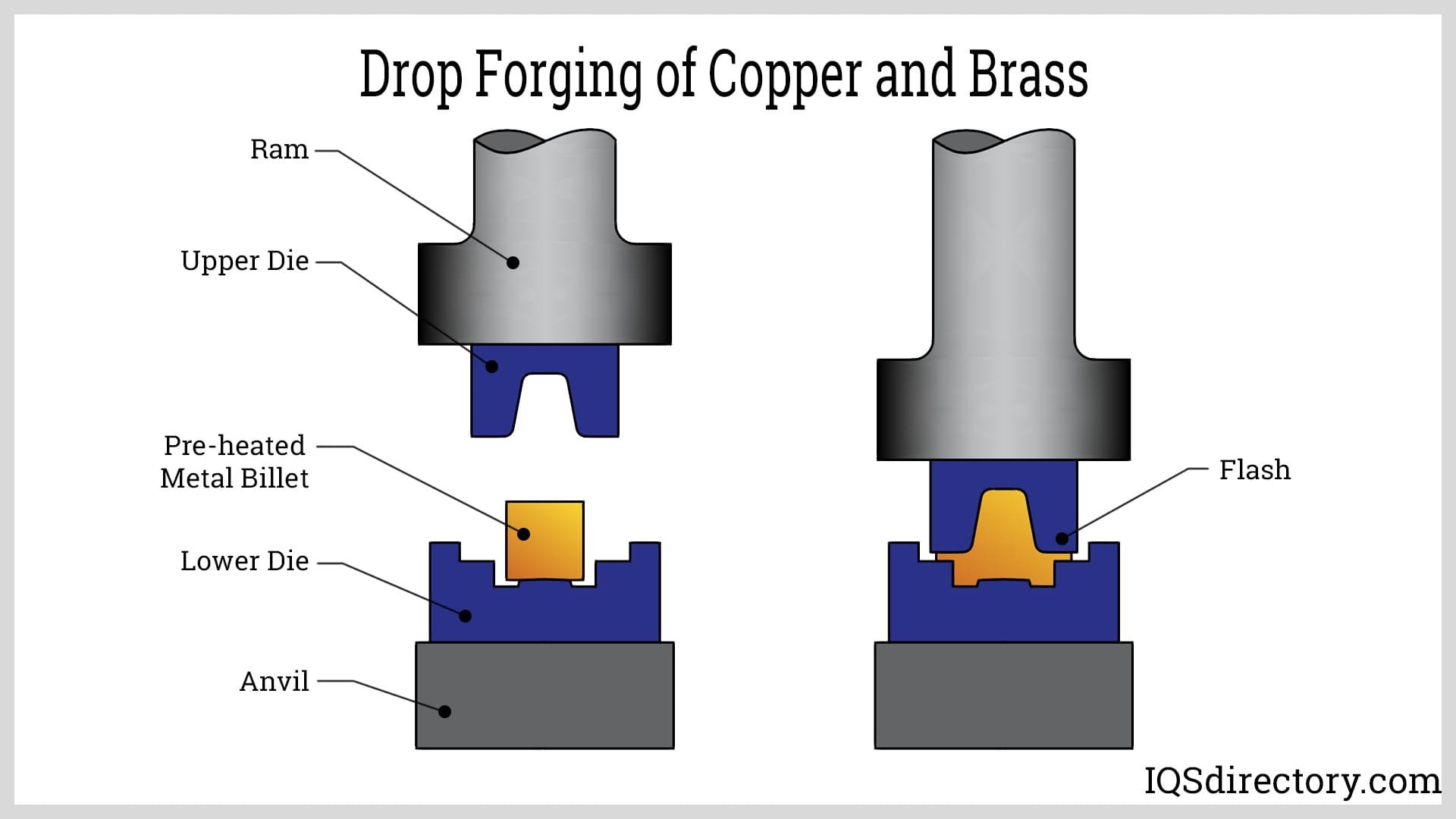
Materials a Blacksmith Can Work With
Steel, Copper, Brass, Aluminium
and Why They Behave the Way They Do
One of the biggest shifts in a blacksmith’s learning journey is realising that forging isn’t just about hitting hot metal — it’s about understanding how different materials respond to heat, force, and time.
Two pieces of metal can look identical at room temperature and behave completely differently once they’re hot. Some steels will harden simply by cooling in air. Others require oil or water. Copper gets harder the more you work it, then suddenly becomes soft again when heated. Aluminium gives almost no visual warning before it fails.
This article explains what materials blacksmiths can work with, and more importantly, why they behave the way they do, so your forging decisions start to feel intentional rather than trial-and-error.
The Core Principle: Heat, Structure, and Atomic Movement
All forging behaviour comes down to how atoms move and rearrange.
When metal is heated, its atomic structure becomes more flexible. Atoms can slide, rotate, and reposition under force. When the metal cools, that structure “freezes” into whatever arrangement it was in at that moment.
Hardening, softening, cracking, and toughness are all consequences of:
how freely atoms can move when hot
how suddenly that movement is stopped
and what other elements are present in the metal
Steel behaves the way it does because carbon interferes with iron’s crystal structure. Copper behaves differently because it has no such mechanism. Aluminium behaves differently again because of how quickly it conducts heat and how its alloys are formulated.
Once you understand this, forging starts to make sense.
Steel: The Foundation of Blacksmithing
Steel is the material most people associate with blacksmithing, and for good reason. It is strong, widely available, and uniquely adaptable. Its defining feature is carbon dissolved within iron, and that carbon is responsible for almost all of steel’s interesting behaviour.
When steel is heated to a critical temperature, its internal structure changes into a form that can dissolve carbon more freely. How that structure is allowed to cool determines whether the steel becomes soft, hard, tough, or brittle.
Mild (Low-Carbon) Steel
Mild steel contains very little carbon. Because of this, it cannot form a hard structure, no matter how quickly it is cooled.
When you heat mild steel and allow it to cool — even if you quench it in water — the internal structure simply relaxes back into a soft, workable state. This makes it extremely forgiving and ideal for learning hammer control, heat management, and basic shaping.
That forgiving nature is why mild steel is so often recommended for beginners. It allows mistakes without permanent consequences.
Medium-Carbon Steel
Medium-carbon steel sits in the middle ground. It contains enough carbon to harden, but not so much that it becomes extremely brittle.
When heated and cooled correctly, medium-carbon steels can become strong and wear-resistant while still retaining toughness. However, they demand more control. Cooling too quickly can cause cracking, while cooling too slowly may prevent proper hardening.
This category includes many practical tool steels and mechanical components, and it is often where blacksmiths begin learning controlled heat treatment.
High-Carbon Steel
High-carbon steel contains enough carbon to harden aggressively. This makes it ideal for cutting tools, springs, and blades — but also makes it unforgiving.
When quenched, high-carbon steel undergoes a dramatic structural change that traps carbon in place, forming a very hard but stressed structure. If that stress isn’t relieved through tempering, the steel can crack or fail suddenly.
This is why high-carbon steel rewards precision and punishes guesswork. It is powerful, but it demands respect.
Why Some Steels Harden in Air and Others Need Quenching
One of the most confusing things for beginners is discovering that not all steels require water or oil to harden.
The reason lies in alloying elements.
Some steels contain additional elements such as chromium, molybdenum, or vanadium. These slow the movement of atoms as the steel cools. As a result, even slow cooling in still air is fast enough to lock the structure into a hardened state.
Other steels lack these elements and require much faster cooling — oil or water — to achieve the same effect.
This is why quenching medium or high-carbon steel in water can be dangerous, while an air-hardening steel can safely harden on the bench without any liquid at all.
Work Hardening: A Completely Different Mechanism
Not all metals harden because of heat.
Some metals harden because of mechanical deformation.
When you hammer certain metals at low or moderate temperatures, you distort their crystal structure. As that distortion accumulates, atomic movement becomes restricted, and the metal becomes harder — but also more brittle.
This process is called work hardening, and it behaves very differently from heat hardening.
Copper: A Perfect Teacher Material
Copper does not harden through quenching. Cooling speed has almost no effect on its hardness.
Instead, copper hardens as you hammer it. Each blow distorts the structure slightly until it becomes stiff and resistant. Heating copper allows that structure to relax, returning it to a soft state.
This creates a repeating cycle:
hammer → harden → heat → soften → repeat.
Because this behaviour is predictable and reversible, copper is an excellent material for learning how metal responds to force and heat without the risks associated with steel hardening.
Brass: Stronger, Less Forgiving Copper
Brass is primarily copper with zinc added. That zinc increases strength but reduces forgiveness.
Like copper, brass work hardens as it is hammered. However, it reaches its brittle state much faster and is more prone to cracking if not annealed regularly.
Brass also introduces a safety concern: overheating can release zinc fumes, which are hazardous if inhaled. This makes good ventilation essential when working brass.
Forged carefully, brass is beautiful and strong. Forged carelessly, it fails abruptly.
Aluminium: Deceptively Difficult
Aluminium is often assumed to be beginner-friendly because it is lightweight and common. In reality, it is one of the least forgiving forging materials.
Aluminium does not change colour visibly as it heats, so it offers almost no visual feedback. It also has a narrow working temperature range and melts suddenly when overheated.
Many aluminium alloys also work harden rapidly, becoming brittle without obvious warning. For these reasons, aluminium is usually better avoided until a smith has good heat awareness and material understanding.
Tin: Why It’s Rarely Forged
Tin has a very low melting point and minimal structural strength. It does not respond well to forging and offers little educational value compared to other metals.
It is far more commonly used in casting or alloyed with copper to create bronze.
Why Quenching Works for Steel — and Not for Other Metals
Steel hardens because carbon becomes trapped inside a rapidly frozen structure. Non-ferrous metals lack that mechanism.
Quenching copper, brass, or aluminium simply cools them. It does not lock their structure into a harder form. Any hardness they gain comes from deformation, not temperature change.
Understanding this distinction prevents many beginner mistakes.
Choosing Materials as a Beginner
Early learning benefits from materials that are:
predictable
forgiving
safe
Mild steel and copper meet those criteria well. High-carbon steel and brass can be introduced gradually. Aluminium and unknown scrap alloys are best left until experience grows.
Final Thought
Blacksmithing becomes dramatically more enjoyable once metal stops feeling mysterious.
When you understand why materials behave the way they do, forging stops being about fighting resistance and starts becoming a conversation between heat, force, and structure.
That understanding is what turns repetition into skill.